At AM Consultancy we specialize in auditing, consulting, and implementation of Good Manufacturing Practices in the Pharmaceutical and Cosmetic Industry.
We help our customer solving their quality risks, design efficient and effective quality systems as a part of quality assurance, manufacturing, and regulatory requirements. We ensure that not only the appropriate quality standards are in place, but also the most efficient processes to increase productivity and product quality.
By Audit we evaluate compliance with the objectives of the established Quality System and Good Manufacturing Practices (GMP) guidelines. We believe in your product quality, putting ‘Quality Compliance from Health to Beauty’ in place.
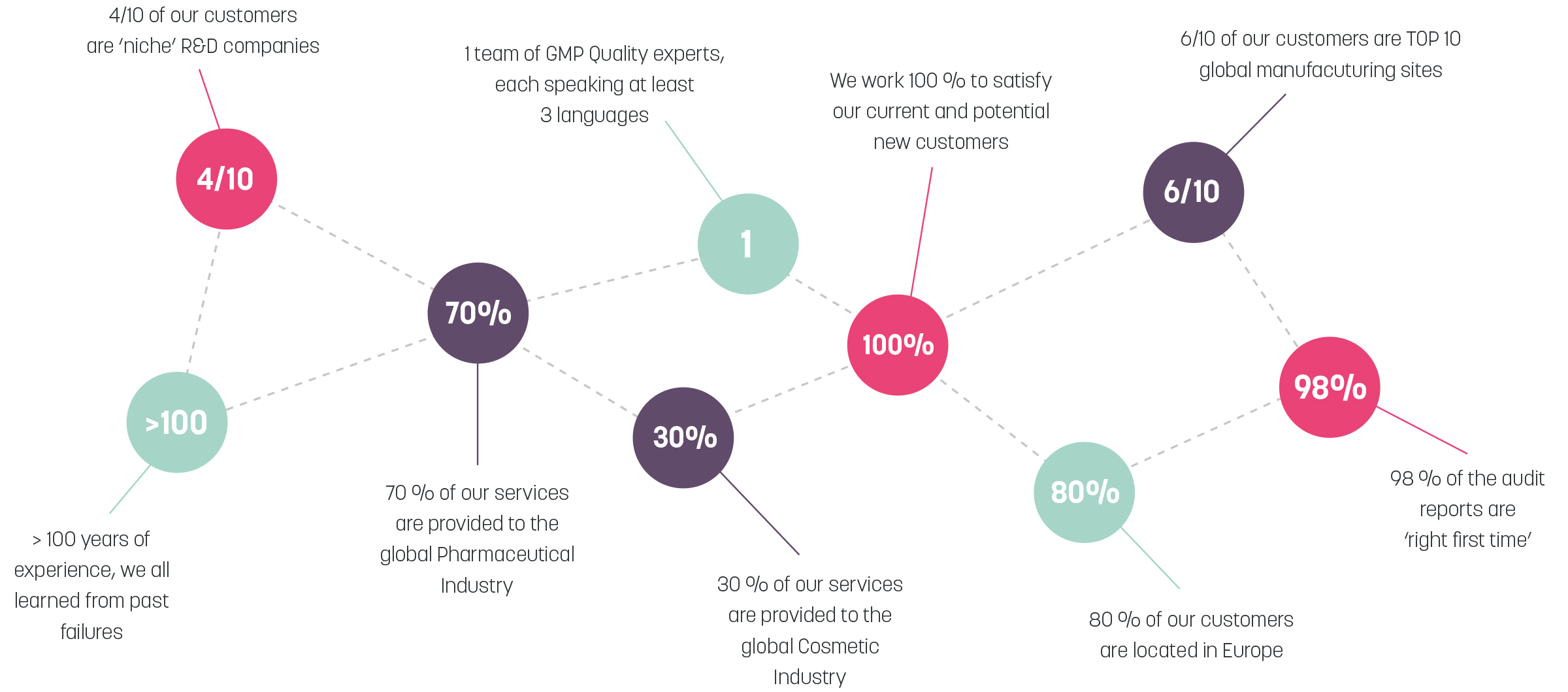
AM Consultancy is a Belgian based company, originally founded as a Pharmaceutical Compliance Consultancy office, the company expanded the scope to the Cosmetic Industry in 2010. AM Consultancy offers services to the primary and secondary Pharmaceutical and Cosmetic Industries within Europe including third-party Audits. On customer requirement, Global Audits are served as well.
Audit activities are performed by the owner of the company Anne Marie (Annemie) Lambert or other senior expert partners.
She started consultancy and audit activities after a career of 20 years+ in Quality Control, Manufacturing and Quality Assurance Management roles in the Pharmaceutical Industry, from R&D to Operations.
She holds a degree in clinical chemistry-microbiology and became a IRCA certified Lead Auditor in 2010. During her career she hosted several European authority, FDA, Anvisa and other regulatory GMP Inspection Audits. Specialized in GMPs for Sterile Medicinal Products.
Proud member of ECA GMP Academy and Board member of Cosmetics Consultants Europe.
Pharmaceutical GMP Audits
Any manufacturer of medicines intended for the EU market, no matter where in the world located, must comply with GMP. Good Manufacturing Practice (GMP) describes the minimum quality standard that a pharmaceutical product manufacturer must meet in their production processes. GMP requires that medicines are of consistent high quality, are appropriate for their intended use and meet the requirements of the marketing authorization or clinical trial authorization.
AM Consultancy supports national and global life science companies from top 10 pharma organizations to emerging start-ups. AM Consultancy assist in the review and evaluation of GMP compliance, performing quality audits at pharmaceutical manufacturing sites.
Cosmetic GMP Audits
One of the main purposes of the European Cosmetic Products Regulation (EC)No. 1223/2009 is to improve the safety of cosmetic products made available to the public of the European market.
Adherence to ISO 22716 ‘Cosmetics- Good Manufacturing Practices (GMP)’ guarantee the production of a cosmetic product that meets defined product characteristics and the requirements established in the Product Information File (PIF), and thus put product on the market that remains safe under normal shipping and usage conditions. AM Consultancy offers audit and GMP compliance services to the cosmetic and personal care industry.

Consultancy activities
Our (inter)national relations






















